Rare and Orphan Disease Focused
Experienced Management Team
Rare Pediatric Designation and US/EU Orphan Disease Designation
Targeting Approval for QRX003 in Netherton Syndrome in 2027
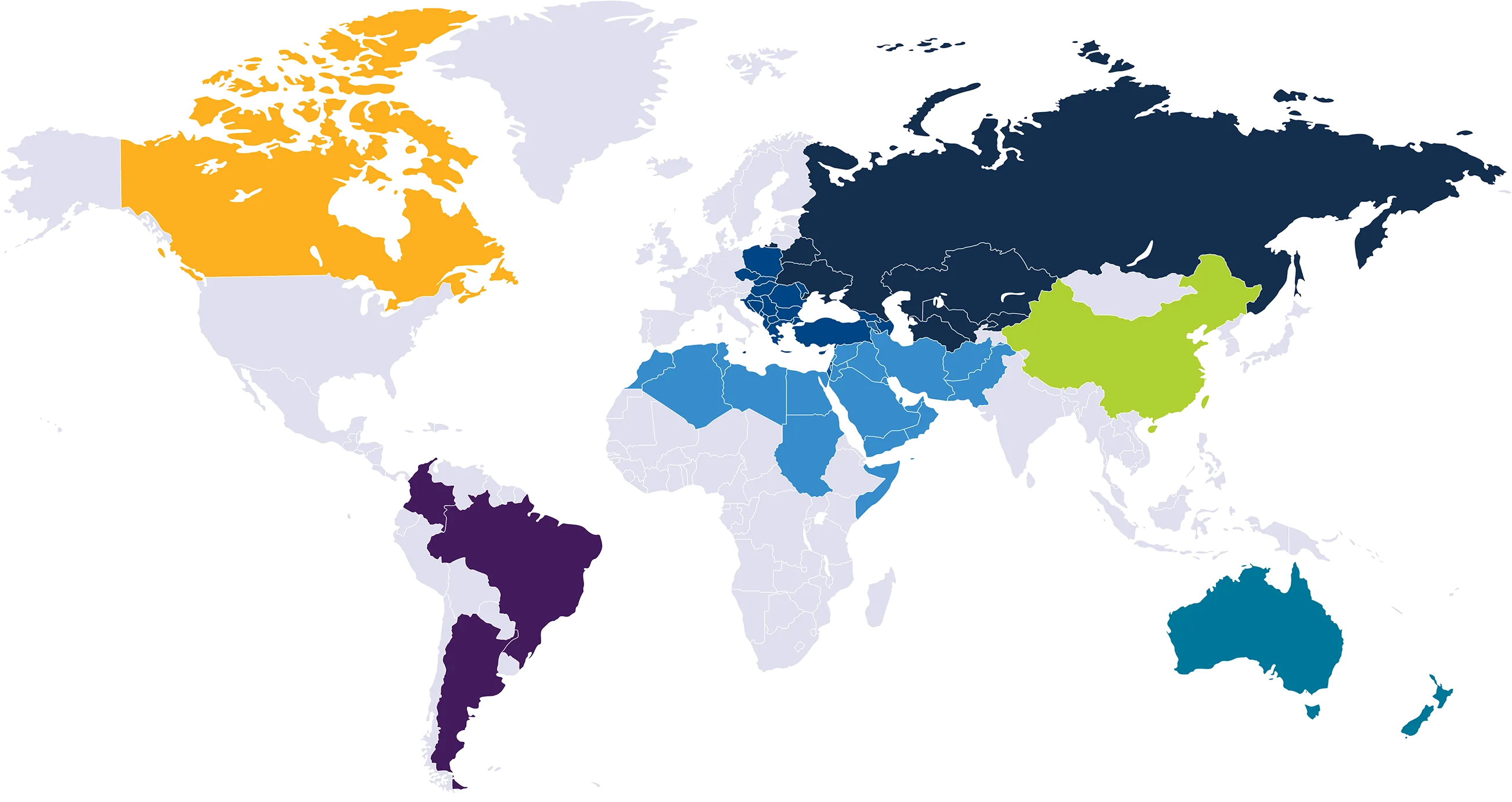
Internal commercial infrastructure planned US, Western Europe and Japanese markets. Marketing partnerships established spanning 61 countries
Strong KOL support
Quoin Pharmaceuticals Ltd. (NASDAQ: QNRX) is a clinical stage specialty pharmaceutical company focused on the development and commercialization of therapeutic drug products that treat rare and orphan diseases. Our mission is very simple; to bring hope where there is currently none. We are totally committed to addressing critical, unmet medical needs for patients, their families, communities and care teams.
Quoin’s innovative pipeline comprises products that collectively have the potential to target a broad number of rare and orphan indications, including Netherton Syndrome, Peeling Skin Syndrome, Ichthyosis, Microcystic Lymphatic Malformations, Venous Malformations, Angiofibromas, Scleroderma, and other additional as of yet undisclosed indications. The company plans to establish a commercial sales infrastructure to support product launches in the US, Western Europe and Japan. In addition, Quoin has entered into nine separate strategic commercial agreements spanning 61 countries including Canada, Australia, New Zealand, the Middle East, China, Hong Kong, Taiwan, Central and Eastern Europe, the Baltic States and several countries in Latin America.
We are dedicated to finding unique solutions for rare and orphan indications and will continue to expand our pipeline of products to address the unmet needs of underserved and neglected patient populations.
Our History
Our Team
Executive Officers
Board of Directors
Scientific Advisors
Dr. Amy Paller
Professor Alan Irvine
Professor Jemima Mellerio
Our Partners