Pipeline
Quoin’s team is relentlessly focused on developing and commercializing innovative treatments for rare and orphan diseases, for which none exists today. Our pipeline comprises three unique products that collectively have the potential to target a broad number of indications, including Netherton Syndrome, Scleroderma, Peeling Skin Syndrome, Ichthyosis, Microcystic Lymphatic Malformations, Angiofibroma, and others. We are committed to continuously expanding our pipeline of products to target additional rare and orphan diseases.
Therapeutic Development Pipeline
| Product Candidate | Indication | Pre-Clinical | Phase 1 | Phase 2 | Phase 3 | Approval | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| QRX003 |
|
|
||||||||||||
| QRX008 |
|
|
||||||||||||
| QRX009 |
|
|
* Pivotal Clinical studies to commence 2026
** Clinical trial initiated
*** Clinical testing to commence 2026
Quoin Pharmaceuticals launched NETHERTON NOW, a campaign dedicated to raising awareness, fostering advocacy, and supporting patients and families affected by Netherton Syndrome.
VISIT THE WEBSITE: nethertonnow.com
Other Target Indications
Peeling Skin Syndrome
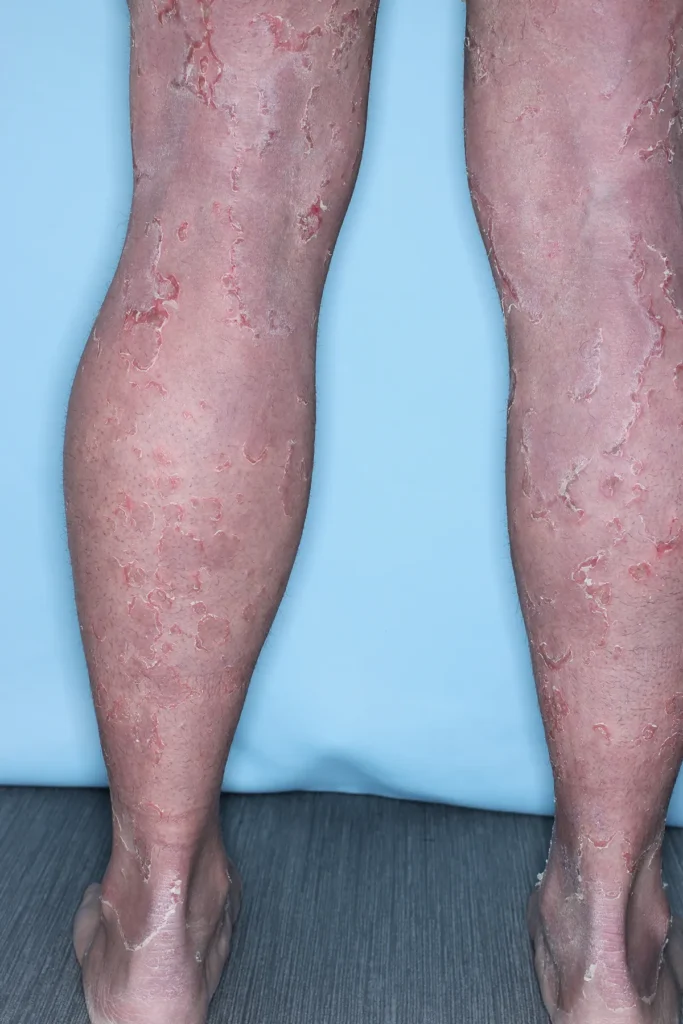
Peeling Skin Syndrome (PSS) is a rare, genetic skin disorder characterized by painless, continual skin peeling due to a separation of the stratum corneum from the underlying layers of the Epidermis. PSS may be generalized, affecting skin over the patient’s entire body, or localized, affecting only the skin of the hands and feet. Generalized PSS can be categorized as non-inflammatory, and less severe, or inflammatory, which is typically more severe and involves other organ systems. There is currently no approved treatment for PSS, and patients manage symptoms using over-the-counter emollients.
Palmoplantar Keratoderma
Palmoplantar Keratoderma (PPK) is a group of rare skin disorders with symptoms including thickened skin on the palms of the hands and the soles of the feet, fragile and blistering skin, and excessive perspiration. In its rarer forms, PPK can affect organs other than the skin. Genetic PPKs result in keratin abnormalities in patients.
SAM Syndrome
SAM Syndrome, also known as severe skin dermatitis, multiple allergies and metabolic wasting, is a life-threatening rare disease caused by mutations in the desmoglein 1 (DSG1) and desmoplakin (DSP) genes. A compromised skin barrier results in a predisposition to allergies, and other symptoms include severe pruritus, palmoplantar keratoderma, pustular psoriasis and excessive sweating. Quoin is pursuing the development of QRX003 as a potential treatment for SAM Syndrome.
Scleroderma
Scleroderma is a rare, autoimmune disease that affects connective tissue of the skin, blood vessels, internal organs and digestive tract. Symptoms of scleroderma include thickening and tightening of the skin, and inflammation that leads to problems in other organs including the lungs, heart and kidneys. There is currently no approved treatment or cure for scleroderma.

Resources
FIRST Skin Foundation for Ichthyosis and related skin types
Supports patients and families affected by Ichthyosis.
NORD National Organization for Rare Disorders
NORD is leading the fight to improve the lives of patients with rare diseases.
Genetic and Rare Diseases (GARD) Information Center
GARD is a program of the National Institutes of Health (NIH) that provides free access to reliable, easy to understand information about genetic and rare diseases.
Molecular Diagnostic Testing for Netherton Syndrome
Gabriele Richard, MD Associate Professor, Department of Dermatology and Cutaneous Biology and Department of Medicine / Division of Medical Genetics Thomas Jefferson University
Ichthyosis Support Group
Committed to the ongoing provision of an information network and support structure for individuals and families affected by ichthyosis.
Expanded Access Policy
We believe that the goal of making treatments available to the patient community is most effectively achieved through rigorous science and well-designed clinical studies followed by regulatory approval. The best way to access an investigational product, i.e., a treatment being studied that has not received regulatory marketing approval, is through participation in a clinical trial. In certain circumstances, we may allow access to people with serious or life-threatening diseases to gain early access to an investigational product outside of a clinical trial when no satisfactory alternative treatment is available. This is called Expanded Access, also referred to as compassionate use.
This policy describes criteria that we will consider to determine if one of our investigational products can be made available for patients outside of clinical study, specifically when the product is not approved for any use in the country where the treating physician is requesting pre-approval access. This policy also applies during the period between regulatory approval of an investigational product and its commercial availability in that country. Criteria that we will consider include:
- The patient is suffering from a serious or immediately life-threatening disease or condition.
- The patient has undergone appropriate standard treatments without success and no comparable or satisfactory alternative treatment is available or exists to treat the disease or condition.
- The patient is not eligible for, or is otherwise unable to participate in, any ongoing clinical study of the investigational product, including due to geographic limitations.
- There is sufficient scientific evidence to suggest that the patient may benefit from the investigational product and that the potential benefit outweighs the known or anticipated risks.
- The investigational product is under active development for the requested disease indication.
- Sufficient evidence exists to suggest the investigational product has an acceptable safety profile for the intended patient population.
- Provision of the investigational product will not interfere with or compromise the ongoing clinical development program.
- Adequate supply of the investigational product exists.
If expanded access is granted, the treating physicians who are properly licensed and qualified must:
- Agree in writing to comply with all applicable country-specific legal and regulatory requirements related to the provision of investigational products under Expanded Access.
- Obtain the necessary approvals from relevant regulatory authorities, ethics committees or institutional review boards (IRBs) and import license for investigational product.
- Ensure the informed consent of the patient is obtained in accordance with applicable laws and regulations.
- Monitor the patient appropriately and fulfill all safety reporting obligations.
Review Process
QUOIN is committed to evaluating all Expanded Access requests in a fair and equitable manner. All requests must be submitted by the patient’s treating physician. QUOIN may require additional information in order to fully assess each request. The Sponsor will acknowledge receipt of expanded access requests and make reasonable efforts to respond in a timely manner. Every request will receive careful consideration; however, access to QUOIN’s investigational therapies is not guaranteed, as approval decisions are made at the sole discretion of the company.
Physicians seeking Expanded Access for patients with no viable alternative treatment options should submit their requests to: expandedaccess@quoinpharma.com. For information on QUOIN’s ongoing investigational studies, please visit: https://quoinpharma.com/pipeline/